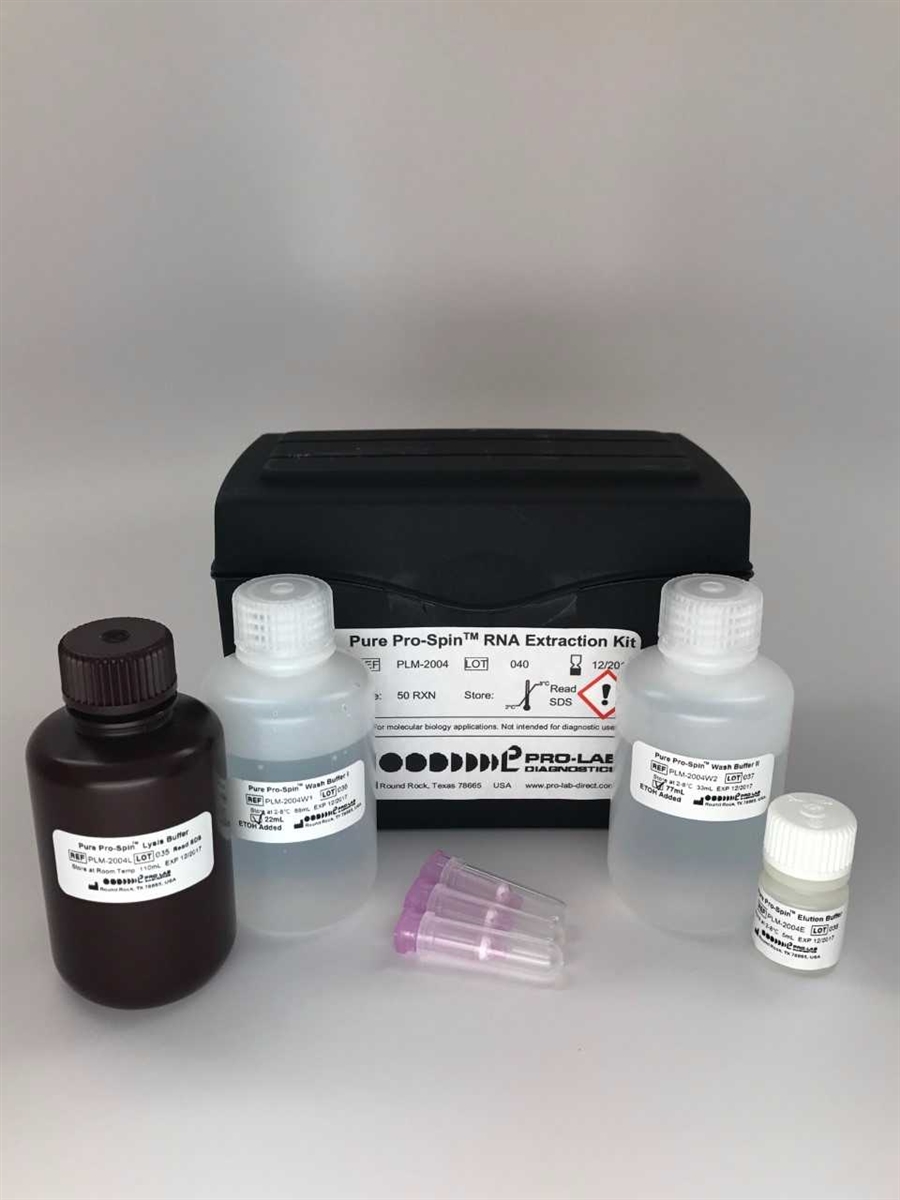
Product & Regulatory Information
Regulatory status: For Research Use Only. Not intended for the diagnosis, prevention, or treatment of disease. Source: IFU PLM-2004 ver-20180706.
Principle: Total RNA isolation via silica-membrane spin column. Lysis buffer denatures and lyses cells, inactivating RNases and releasing RNA. RNA binds selectively to the silica gel membrane under chaotropic buffer conditions. Contaminants are removed by wash buffers. Pure RNA is eluted in low-salt buffer. [Corroborated — product page description + established silica-membrane chemistry]
RNase QC: All buffer components are quality control checked for RNase contamination. [Claimed — product page] Per-run QC requirements (controls, frequency) — confirm from IFU when received.
Hazard information: [FLAG — SDS not yet received. GHS pictograms, signal word, hazard statements, PPE requirements, and disposal classification to be added upon SDS acquisition. Request from info@pro-lab.com.]
IFU and SDS: Instructions for use and safety data sheet available on request. Contact info@pro-lab.com or download from the product page at pro-lab-direct.com/product-p/plm-2004.htm.